Biopreparedness
Item set
Title
Biopreparedness
Items
64 items
-
Andre Kalil, MD, MPH
In February 2020, Andre Kalil, MD, MPH, led a team at Nebraska Medicine in enrolling some of the first COVID-19 patients in a National Institute of Health clinical trial to study remdesivir as a treatment for the disease. The study confirmed remdesivir as an effective treatment and was approved for emergency use in May 2020. The drug received full FDA approval in October 2020 and continues to be a form of treatment for patients with severe COVID-19 conditions. -
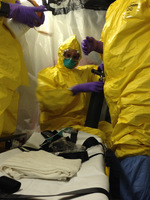
Biocontainment Personal Protective Equipment , c. 2014
In July 2014, representatives from the U.S. State Department visited Nebraska Medicine to evaluate the capabilities of the Nebraska Biocontainment Unit (NBU). They determined the unit was well prepared to care for patients. This approval led to the unit’s first activation in September 2014 to treat patients with the most dangerous infectious disease then known: the Ebola Virus Disease. Biocontainment suits and personal protective equipment (PPE) like the one displayed here were worn by the NBU team during early activation to transport and receive patients. The suit consists of multiple layers put on in a specific order to seal in and protect the wearer. Discover more about donning and doffing biocontainment PPE via the interactive screen. -
Biocontainment Personal Protective Equipment, c. 2014
In early September, the Nebraska Biocontainment Unit (NBU) successfully treated its first patient, Rick Sacra, MD. Dr. Sacra contracted Ebola while treating patients in Liberia. He was discharged from the NBU later that month. In early October, the unit received and treated a second patient who was discharged later that month after being declared Ebola-free. While the biocontainment suits were used during the early stages of transporting and treating patients, the NBU team transitioned to the high-level PPE displayed here once the patient was settled in the secure unit. -
Biocontainment Personal Protective Equipment, c. 2020
The Nebraska Biocontainment Unit (NBU) activated for the second time in its history in the early days of the COVID-19 pandemic. The National Quarantine Unit activated for the first time as well, caring for Americans returning from Wuhan, China, in February 2020, and receiving patients from the Diamond Princess cruise ship shortly after. The Powered Air Purifying Respirator level Personal Protective Equipment (PAPR PPE) on display was worn in the NBU to treat patients with severe COVID-19 symptoms. -
Biological and Chemical Agent Guide, April 2005
After the terror attacks in 2001, the Centers for Disease Control and Prevention (CDC) prepared binders with information about possible biological and chemical agents that could be used in an attack. The UNMC Center for Preparedness Education duplicated binders like this to share with public health officials across Nebraska who were developing bioterrorism protocols for their districts. -
Board of Regents NBU Resolution, January 30, 2015
The Nebraska Biocontainment Unit (NBU) received honors across the country for their care of Ebola patients in 2014. Some of the accolades included: Special recognition by the White House and then-President Barack Obama The Omaha World-Herald’s Midlander of the Year Award In addition, the NBU team was honored by the Board of Regents in this Resolution applauding their courage, exemplary leadership, and thanking them for their service. -
Board of Regents NBU Resolution, January 30, 2015
The Nebraska Biocontainment Unit (NBU) received honors across the country for their care of Ebola patients in 2014. Some of the accolades included: Special recognition by the White House and then-President Barack Obama The Omaha World-Herald’s Midlander of the Year Award In addition, the NBU team was honored by the Board of Regents in this Resolution applauding their courage, exemplary leadership, and thanking them for their service. -
Board of Regents NBU Resolution, January 30, 2015
The Nebraska Biocontainment Unit (NBU) received honors across the country for their care of Ebola patients in 2014. Some of the accolades included: Special recognition by the White House and then-President Barack Obama The Omaha World-Herald’s Midlander of the Year Award In addition, the NBU team was honored by the Board of Regents in this Resolution applauding their courage, exemplary leadership, and thanking them for their service. -
Camp Ashland Entrance, 2020
On February 7, 2020, UNMC and Nebraska Medicine answered the national call to support 57 Americans returning from Wuhan, China, in need of quarantine and care during the initial outbreak of COVID-19. Upon arrival, patients were safely housed at the Nebraska National Guard’s Camp Ashland location for a 14-day quarantine period as mandated by the CDC. By February 20, 2020, all 57 patients were released after two negative COVID tests. -
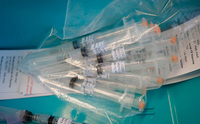
CDC COVID-19 Test Kits, 2020
In a rush to provide testing to health care centers across the country, the Centers for Disease Control and Prevention (CDC) unfortunately released faulty kits, like the ones displayed here. The error was quickly discovered by the labs of UNMC and Nebraska Medicine, who worked with the CDC to remedy the issue. -
CDC COVID-19 Test Kits, 2020
In a rush to provide testing to health care centers across the country, the Centers for Disease Control and Prevention (CDC) unfortunately released faulty kits, like the ones displayed here. The error was quickly discovered by the labs of UNMC and Nebraska Medicine, who worked with the CDC to remedy the issue. -
Center for Preparedness Education Logo, c. 2006
PrepEd operates out of the College of Public Health’s Center for Biosecurity, Biopreparedness, and Emerging Infectious Diseases (CBBEID). -
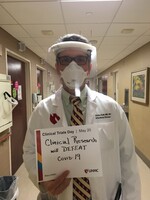
Clinical Research Unit Ribbon Cutting, February 2021
The center’s new Clinical Research Unit gave UNMC and Nebraska Medicine the infrastructure to serve as a host site for a national clinical trial investigating COVID-19 vaccines for adults. -
COVID-19 Diagnostic Testing
Shortly after development of the first CDC testing kits for COVID-19, scientists in public health labs across the U.S. confirmed the tests were faulty. While the Nebraska Public Health Laboratory worked to fix the test kits on hand, M. Jana Broadhurst, MD, PhD, DTM&H, director of the Emerging Pathogens Laboratory, and her team worked with the CDC to develop better and faster diagnostic testing. They eventually created the saliva Polymerase Chain Reaction (PCR) test which is currently on display. -
COVID-19 PCR Saliva Test, November 2020
In the search for effective testing methods for COVID-19, a team in the Emerging Pathogens Laboratory at UNMC developed this novel saliva Polymerase Chain Reaction (PCR) test for fast and accurate screening. Collecting a sample is easy—you just spit in a tube! Clinical trials proved the test to be very effective at detecting COVID-19 in asymptomatic individuals. -
COVID-19 PCR Saliva Test, November 2020
In the search for effective testing methods for COVID-19, a team in the Emerging Pathogens Laboratory at UNMC developed this novel saliva Polymerase Chain Reaction (PCR) test for fast and accurate screening. Collecting a sample is easy—you just spit in a tube! Clinical trials proved the test to be very effective at detecting COVID-19 in asymptomatic individuals. -
COVID-19 Vaccine
On December 11, 2020, the FDA gave Emergency Use Authorization (EUA) for the Pfizer-BioNTech COVID-19 vaccine, and an EUA for the Moderna vaccine following shortly after. UNMC/Nebraska Medicine used the first doses they received to vaccinate frontline health care workers, with vaccination becoming more widely available for the public in February 2021. As the virus continues to evolve, new bivalent booster vaccinations have become the best option to protect people and decrease the spread of COVID-19. -
Diamond Princess Cruise Ship
On February 13, 2020, UNMC/Nebraska Medicine received 13 Americans from the Diamond Princess cruise ship. Two additional passengers were later transferred to the medical center campus and united with their spouses in quarantine. Of the 15 passengers, 13 tested positive for COVID-19. The patients were primarily housed at the National Quarantine Unit on campus, but some were housed in the NBU to receive more intensive care. All patients were released by mid-March after testing negative. -
Doffing COVID-19 PPE
Doffing PPE in the correct order protects the health care worker from exposure and prevents the spread of contaminants and infectious diseases. This interactive demonstrates the proper doffing and sanitization protocols with respect to COVID-19. To interact: Drag and drop PPE from the actor into the dispose area to doff Drag and drop hand sanitizer on the actor to sanitize hands -
Donning COVID-19 PPE
COVID-19 PPE is designed to protect the health care worker from exposure to the respiratory droplets that spread the virus. This activity demonstrates the donning order as well as proper sanitizing steps. To interact: Drag and drop PPE onto the actor to don Drag and drop hand sanitizer on the actor to sanitize hands -
Donning High Level PPE
https://webmedia.unmc.edu/iexcel/apps/PPE2/PPE_Part_2.html -
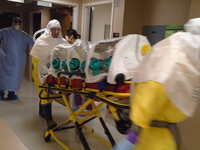
Ebola patient arrival, c. 2014
Biopreparedness efforts at UNMC and Nebraska Medicine were put to the test in 2014 when the Nebraska Biocontainment Unit received and cared for three Ebola patients. -
Edward Jenner, portrait
English physician and scientist Edward Jenner is credited with pioneering the concept of vaccines, having developed an effective inoculation for smallpox in the 1790s. Jenner noticed that milkmaids who had recovered from a milder disease, cowpox, appeared to have a natural immunity to smallpox. In 1796, Jenner tested the theory by inoculating an 8-year-old boy, James Phipps, with a live cowpox specimen. After he recovered, Jenner inoculated James with live smallpox and confirmed the boy was protected.